Simpson, in Organic Structure Determination Using 2-D NMR Spectroscopy (Second Edition), 2012 2.1.3 Solvent Selection Trimethylsilyl trifluoromethanesulfonate μ-wave N, N, Ń, Ń-tetramethylazodicarboxamide TMSOTf Tonsil actisil FF, commercial bentonitic clay TBABĢ-(1 H-benzotriazolyl)-1,1,3,3-tetramethyluronium TFA Using flow charts for paints the type of resin employed can be identified, and in some cases, for example, alkyl and phenolic resins, quantitative determination of resins can be made from the relative areas of the spectrum peaks.ġ3C NMR provides better results than 1H NMR for the resolution of peaks obtained from high molecular mass and very viscous polymers, but the instrumentation required is more expensive and a larger amount of sample and longer analysis times are involved.Ģ,3-dichloro-5,6-dicyano-1,4-benzoquinone DHPġ-ethyl-3-(3-dimethylaminopropyl) carbodiimide Et The NMR spectra of binders provide information about the functional groups present. Just like to add that the Deuterium is also used by the spectrometer as lock signal to add extra frequency stability to the spectrum.Salvador Garrigues, in Encyclopedia of Analytical Science (Third Edition), 2019 Nuclear magnetic resonance spectroscopyġH and 13C nuclear magnetic resonance (NMR) are effective for the characterization of resins in the binders of paints.īinders for NMR analysis must be isolated from the sample and dissolved in deuterated chloroform (a very good solvent for many resins as it produces only small proton impurity peaks and is less expensive than other deuterated solvents). Then, in the integral options, you could exclude the solvent signals.
Why do you think it's better? I would also assume that it is water though. Finally, integrate the region of interest and the solvent peaks will not be taken into account for the integration. I use the CHCl3 peak and calibrate that to 7.26 ppm. You can never get pure 100 % pure D compunds as is it is an isopote of H, if you read the labal of the bottle it will say some like 99.99% CDCl3 the other 0.01 % is CHCl3, and it is the hydrogen from CHCl3 that is the cause of the peak.
#Mestrenova solvent peaks nmr cloroform how to
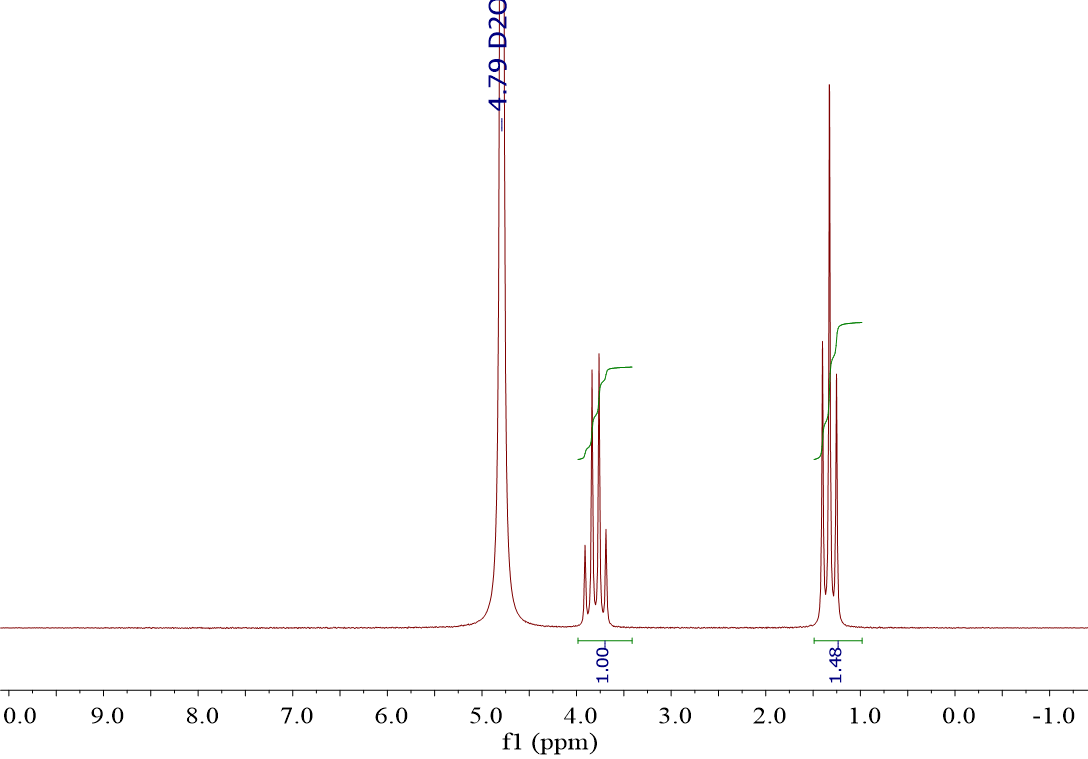
How to integrate CDCl3 (d-chloroform) peak area which is overlapped with other peaks? have used it more times than I can count. It's a really good habit to get into and it also may give you a better idea as to what that peak is.
#Mestrenova solvent peaks nmr cloroform pro
leaders, Yang dismayed by Asian American reaction to Trump, Senator's 'tone deaf' tweet on Lakers, Dodgers slammed, AOC calls Biden's new chief of staff a 'unifying pick', COVID-19 vaccine may have unpleasant side effects, CDC outlines which masks are most effective, Former 'DWTS' pro felt lost after Seacrest breakup, How Trump lost sports as a political strategy, Short lockdown could halt virus spike: Biden adviser, Nancy Pelosi calls on Republicans to 'stop the circus'. First time here? 'Crazy train': GOP lawmaker rips Trump's fraud claims, Disney CEO 'extremely disappointed' in Calif.

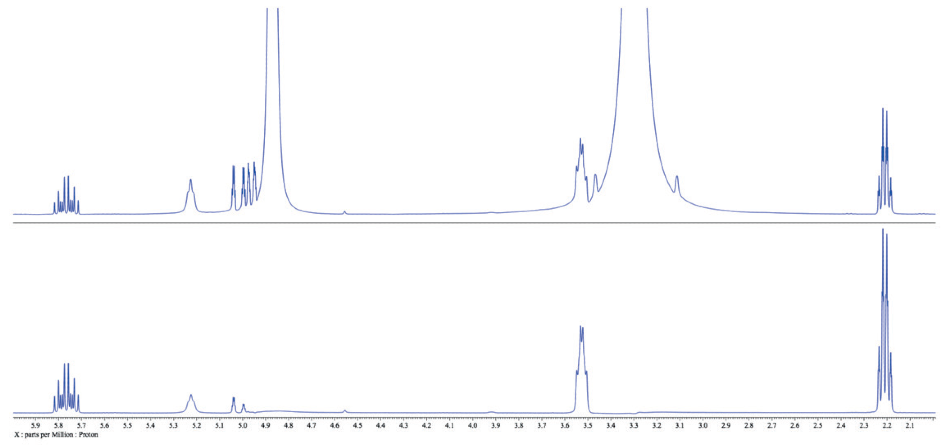
It is used because most compounds will dissolve in it, it is volatile and therefore easy to get rid of, and it is non-reactive and will not exchange its deuterium with protons in the molecule being studied. Water can sometimes be broad this depends on the overall sample concentration, acidity of the solution and relative concentration of other labile protons. This is typical of the appearance of water in $\ce$ is indeed water. It may shift a little bit because of the hydrogen bonding, but i should become more intense. Still have questions? I only use DMSO after I determine a sample is insoluable in CDC元. water peak in CDCl3 for 1H nmr I have run 8 1H NMR spectra of purified compounds in CDCl3 and see a common broad peak at about 1.8 ppm. Tanks! you could just confirm the presence of water by adding some to the sample. I would like to get suggestion about how to integrate the peak area of CDCl3 which is overlapped with other peaks? This method can completely remove the correlation between solute and solvent peaks and thus provides the most accurate quantification in cases of severe overlap and higher order effects.
